What is Iso Propanol?
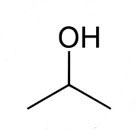
Isopropanol (also known as 2-propanol, Isopropyl alcohol, Propan-2-ol or rubbing alcohol) is a structural isomer of propanol (propyl alcohol) and has the molecular formula of C3H8O. It is a clear, colourless and volatile liquid and has a distinctive strong alcohol or acetone-like odour. This solvent is highly flammable, with a flashpoint of around 11 – 13° C. Isopropanol is soluble in most solvents, including water.
Isopropanol is a common sterilising agent with antibacterial properties which is used in many cleaning applications including hand wipes, disinfectants, hand sanitisers and spray-in-air or ultrasonic products. It also has effective uses in hospitals and medical centres including eliminating skin pathogens.
Solutions of Isopropanol of around 70% are the most effective at disinfection and sterilisation (with a 10 – 40% solution of purified water). This is because when concentrations of alcohol drop below 50%, its sterilising properties decrease at a considerable rate and consequently when this percentage rises to 90%, these properties sustain their disinfection rates.
Technical Properties
Isopropanol and propyl alcohol (1-propanol) are structural isomers of each other and have similar molecules, the difference being their -OH is attached at a different location. It is miscible in water (infinitely), ether, chloroform, acetone and alcohol and chloroform and insoluble in salt solutions.
Technical properties of Isopropanol:
Molecular Formula: C3H8O, CH3CHOHCH3, (CH3)2CHOH
Molecular weight: 60.1 g/mol
Acidity: 16.5ph
Flashpoint: 53°F / 11.7°C
Boiling Point: 180.5°F / 82.3°C
Melting Point: -127.3°F / -89.5°C
Density: 0.786 g/cm3 (20°C)
Refractive index: 1.3772 (20°C)
Log P: -0.16
How is Isopropanol Produced?
The sterilising agent is chemically manufactured using several commercial methods of direct and indirect hydration.
The direct method (and the more modern adaptation) of manufacturing is by reacting very high-quality propene (90-99%) with water with an acidic catalyst, which removes the requirement of sulphuric acid. The two are heated and the vapour produced flows through a reactor that contains sulfonated polystyrene cation ion exchange resins.
The indirect method is a two-step process; step one is reacting propene and sulphuric acid to form sulphate esters and the second step is the hydrolysis of sulfonate esters into isopropanol. Separate reactors are used for the two different steps, and, unlike the direct method, this process can use a lower quality grade of propene.
In both the above methods, the mixture of water and by-products need to go through a distillation process to separate the isopropanol (around 90% purity).
Handling, Storage & Distribution
Storage & Distribution
Isopropanol should be stored in an air-tight storage unit/container in a cool, dry place at room temperature (avoid preserving in hot environments). Isopropyl alcohol can react with oxygen and air over time, forming unstable peroxides which can explode if it is incorrectly stored.
The environment should be well ventilated, and it must be kept away from oxidising agents. Isopropanol should also be protected from direct sunlight, heat and sparks or open flames as it is highly flammable. Containers such as iso tanks made of stainless steel, aluminium or carbon steel are suitable storage units.
A solvent stockist, such as Solventis, would have a bulk petrochemical storage facility to maintain the product with all relevant safety considerations in place.
A bulk solvent exporter would normally distribute this solvent in bulk vessels or tank trucks. For transportation purposes, isopropanol is classed as a flammable liquid with a fire hazard rating of 3 and is a packing group 2.
Isopropanol Health Hazards
Inhalation of Isopropanol can cause irritation and burning sensations in the mouth, throat, eyes and nose. Ingestion can lead to drowsiness, dizziness, increased heart rate, stomach upsets and, in more severe cases, heart problems.
It is advised that eye protection should be always worn when handling isopropanol, if any clothing becomes soaked in the chemical it should be removed immediately and not worn again until clean.
Isopropanol is also known to easily absorb through the skin. Consequently, prolonged, or repeated contact can cause rashes, itching, skin dryness, and redness. Skin contact should be avoided by using protective clothing when handling.
Isopropyl Alcohol Uses
Isopropanol is used as both an intermediate and a solvent in an array of industries, most popularly in the pharmaceutical industry but also in the automotive, laboratory and disinfectant industries.
Solvent
Isopropanol is used as a solvent and cleaning fluid as it is non-toxic compared to other alternatives, for example in consumer uses such as paints, thinners for wood furnishings, inks, dye solutions, cosmetics, and toiletries (lotions, body oils, polishes, shampoos, hair sprays) lubricants, greases, glasses cleaners, and disinfectants.
Intermediate
Isopropanol is used as a chemical intermediate in the manufacture of acetone and its by-products through the process of oxidisation. Other applications include esterification to produce isopropyl acetate and reacting with aluminium metal and titanium tetrachloride to form titanium and aluminium isopropoxide.
Laboratory
In DNA extraction, isopropyl alcohol is added to precipitate DNA, which is then centrifuged to form a pellet to be studied. Therefore, iso propanol has great application in studying the genetic causes of disease to allow for new diagnostics and treatments to be developed. In addition to being used in forensic science, in which DNA extraction is used for determining paternity or sequencing genomes.
Isopropyl alcohol can also be used as a biological specimen preservative with a solution mixture of around 90%. It has benefits over alternatives such as formaldehyde as it is less toxic and thus easier to be used in laboratories.
Disinfectant/Pharmaceutical
In combination with 70% alcohol and a 10 - 40% solution of purified water, isopropanol is an extremely effective steriliser. For this reason, it is found in disinfectant products for homes, rubbing alcohol, hand sanitisers, care products, household cleaning products, and personal care products.
This 70% concentration allows the IPA to permeate the whole cell of microorganisms and cause the proteins in the cell to coagulate or clot. This quickly kills the microorganism.
Water is added to slow the evaporation of the isopropanol, increasing contact time with the surface that is being cleaned and improving the effectiveness of the product. Although concentrations of IPA that are too highly diluted will not be able to permeate the cell wall of microorganisms and therefore be less effective.
Isopropyl alcohol is especially effective for the cleaning of glass surfaces, such as mirrors and windows. The high volatility of the chemical means that it evaporates very quickly, leaving no residue or streaks. As a result, IPA is commonly used in the electronics industry for cleaning specialist parts after manufacture, where no residue must be left for the function of the component.
Additionally, isopropanol is used in medical and pharmaceutical applications, such as laboratory chemicals, and bathing solutions. As a water-drying aid, it can be used to prevent a swimmer's ear (also known as otitis externa).
Automotive
Automotive industry uses of isopropanol include anti-freeze products, adhesives, coating additives and paints. It also plays an important role in making the water component of petrol more soluble so that it does not freeze. Due to its quick-drying and streak-free cleaning, it is also used in the detailing industry, such as for sticker residue removal.
The information above shows that isopropanol has a variety of practical applications and can have significant benefits in our daily lives when it is handled correctly.
Solventis as your Isopropanol Supplier
Solventis is a leading bulk supplier and distributor of isopropanol or IPA in the UK, Europe and globally. As a company with offices in the UK and Belgium, we are proud to be able to offer a personal and efficient service, together with highly competitive prices.
As a result of our state-of-the-art facility in Antwerp, Belgium, we can guarantee quality, efficiency, safety, and environmental awareness for our whole range of products that is second to none. Please contact us for more information on our isopropyl alcohol or any other alcohol from our range by requesting a specification or by phone on +44 (0)1483 203224.



